Synthesis and characterization of the arylomycin lipoglycopeptide antibiotics and the crystallographic analysis of their complex with signal peptidase
J. Liu, C. Luo, P.A. Smith, J.K. Chin, M.G.P. Page, M. Paetzel, F.E. Romesberg, J. Am. Chem. Soc. (2011) 133:17869-17877.
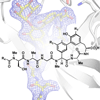 We report the first total synthesis of a lipoglycopeptide member of the arylomycin family of natural products, and we demonstrate that it effectively penetrates Gram-negative bacteria and is limited by the same resistance mechanism as other members of the arylomycin class of antibiotics (i.e. mutation in the target protein signal peptidase (SPase)). Unlike the other arylomycins, the lipoglycopeptides are glycosylated, and the structural data reveal that this sugar is most solvent exposed when in complex with SPase.
We report the first total synthesis of a lipoglycopeptide member of the arylomycin family of natural products, and we demonstrate that it effectively penetrates Gram-negative bacteria and is limited by the same resistance mechanism as other members of the arylomycin class of antibiotics (i.e. mutation in the target protein signal peptidase (SPase)). Unlike the other arylomycins, the lipoglycopeptides are glycosylated, and the structural data reveal that this sugar is most solvent exposed when in complex with SPase.